
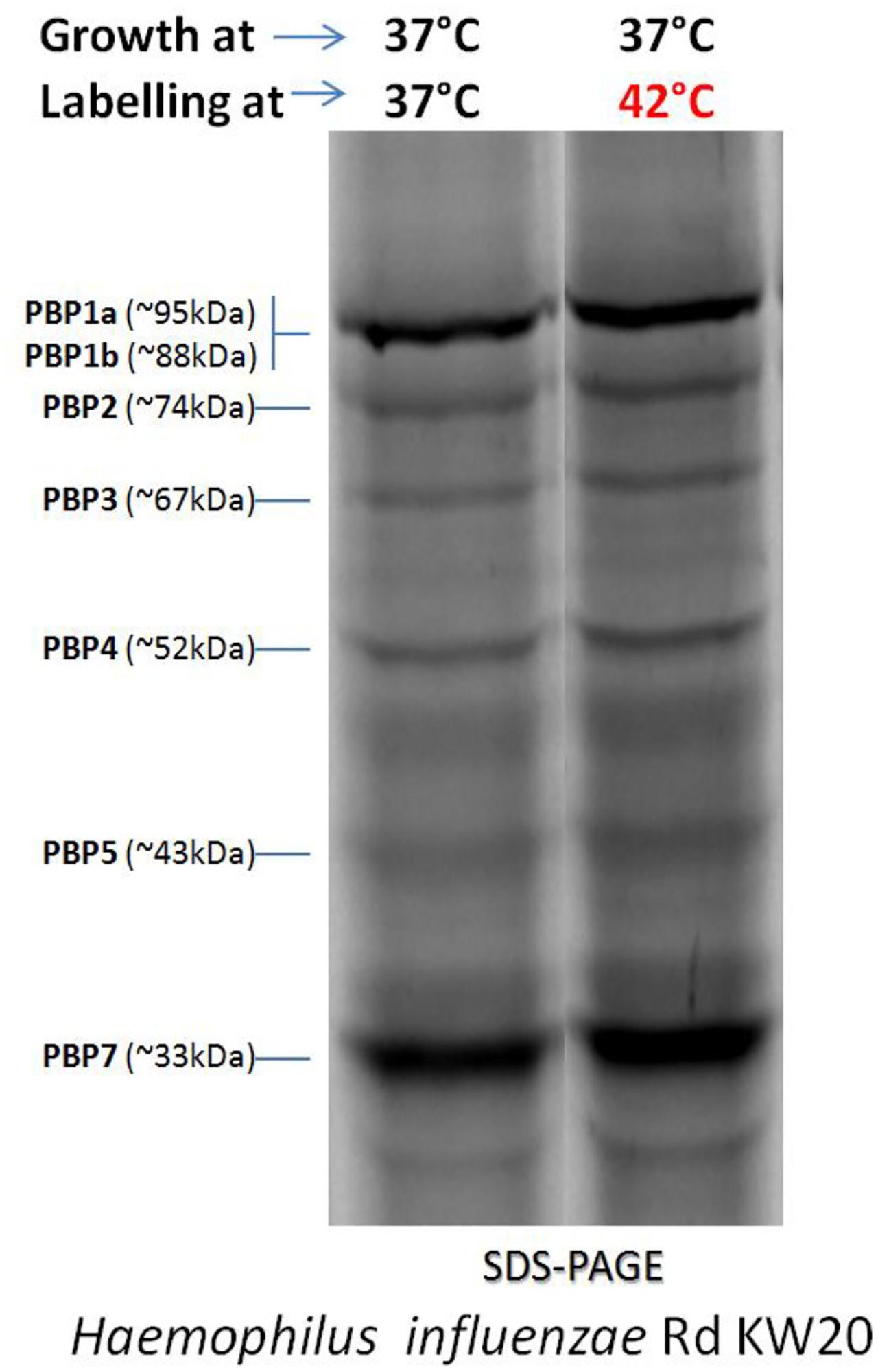
Our analyses indicate that the sequential action of PBP2 and PBP3 drives changes in peptidoglycan organization that are essential for the polarized division of these obligate intracellular bacteria. We have solved the crystal structure of a soluble form of PBP3 (PBP3(57-577)) at 2. PBP3 is mainly periplasmic, with a 23 residues cytoplasmic tail and a single transmembrane helix. Consistent with their distinct roles in polarized division, peptidoglycan organization is different in cells treated with PBP2 and PBP3-specific inhibitors. In Escherichia coli, penicillin-binding protein 3 (PBP3), also known as FtsI, is a central component of the divisome, catalyzing cross-linking of the cell wall peptidoglycan during cell division. The stability of penicillin-binding protein 3 (PBP3), a cell septum synthesizing protein, was analyzed at different incubation temperatures in three. Cells treated with inhibitors that prevent peptidoglycan synthesis or peptidoglycan crosslinking by penicillin-binding protein 2 (PBP2) are unable to initiate polarized division, while cells treated with inhibitors that prevent peptidoglycan crosslinking by penicillin-binding protein 3 (PBP3/FtsI) initiate polarized division, but the process arrests at an early stage of daughter cell growth. tuberculosis H37Rv genome, based on sequence sis by recruitment of penicillin-binding protein 3 (PBP3, or alignments (Fig. During division, peptidoglycan assembles at the pole of dividing Chlamydia trachomatis cells where daughter cell formation occurs, and peptidoglycan regulates at least two distinct steps in the polarized division of Chlamydia trachomatis and Chlamydia muridarum. In However, we feel that pbp3 could arguably be encom- addition to its role in stabilizing the FtsZ ring, FtsW has passed by nucleotides 24268052425046 of the been suggested to facilitate septal peptidoglycan synthe- M. Localization of PBPs to the septum was found to be dependent on FtsZ, but the GFP-PBP fluorescence patterns were not detectably altered in the absence of MreB or Mbl.Chlamydia trachomatis serovar L2 and Chlamydia muridarum, which do not express FtsZ, undergo polarized cell division. The results implicate PBP3, PBP5 and PBP4a, and possibly PBP4, in lateral wall growth. This suggests that cell wall synthesis is not dispersed but occurs at specific places along the lateral cell wall. All other PBPs localized to both the septum and the lateral cell wall, sometimes with irregular distribution along the lateral wall or a preference for the septum. PBP3 and PBP4a localized specifically to the lateral wall, in distinct foci, whereas PBP1 and PBP2b localized specifically to the septum. Penicillin-binding proteins (PBPs), membrane-associated macromolecules which play key roles in the cell wall synthesis process, have been exploited for over 70 years as the targets of the highly successful -lactam antibiotics. The localization of these fusions was studied in a wild-type background as well as in strains deficient in FtsZ, MreB or Mbl. To gain further insights into the topological control of cell wall synthesis in bacteria, we have constructed green fluorescent protein (GFP) fusions to all 11 penicillin-binding proteins (PBPs) expressed during vegetative growth of Bacillus subtilis.

In most non-coccoid bacteria, this shape is also determined by an internal cytoskeleton formed by the actin homologues MreB and/or Mbl.

Bacterial cell shape is determined by a rigid external cell wall.
